
Publications

24. Hyperconjugation Effect in the Conformational Preference of Cyclohexene-Based β-Amino Acids
Jieun Lee, Minseok Oh, Seonho Shin, Younghun Kim, Philjae Kang, Young Kee Kang, and Soo Hyuk Choi
The Journal of Organic Chemistry, ASAP

23. Controlled packing of metal–peptide superhelices with β-peptide foldamers
Ingyu Han, Ha-Jin Lee, and Soo Hyuk Choi,
Organic & Biomolecular Chemistry, 2026, 24, 1600-1604

Ingyu Han, Ha-Jin Lee, Ilia A. Guzei, and Soo Hyuk Choi,
Nature Communications. 2025, 16, 9467

Ingyu Han, Chae Na Lim, and Soo Hyuk Choi,
The Journal of Organic Chemistry. 2025, 90, 42, 15099–15103

Nuri Seo, Yujin Kim, Jieun Lee, Jieun Kim, Philjae Kang, Ilia A. Guzei, Ha-Jin Lee, and Soo Hyuk Choi
Organic & Biomolecular Chemistry, 2025,23, 6133-6137

Nuri Seo, Hoyang Son, Yonghan Kim, Ilia A. Guzei, Philjae Kang, and Soo Hyuk Choi,
Organic Letters. 2023, 25, 41, 7497–7501

18. Conformer-Specific Spectroscopy and IR-Induced Isomerization of a Model γ-Peptide: Ac-γ4-Phe-NHMe
Joshua L. Fischer, Karl N. Blodgett, Christopher P. Harrilal, Patrick S. Walsh, Zachary S. Davis, Sunglim Choi, Soo Hyuk Choi, and Timothy S. Zwier,
The Journal of Physical Chemistry A, 2022, 126 (11), 1837-1847

Sunglim Choi, and Soo Hyuk Choi,
Bulletin of the Korean Chemical Society, 2022, 43( 2), 241.
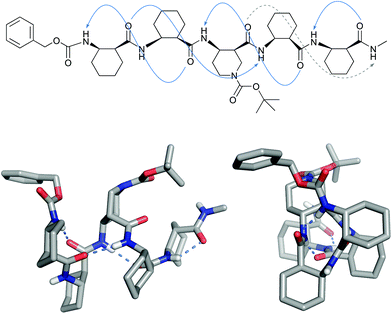
Sunglim Choi, Jihyun Shim, Philjae Kang and Soo Hyuk Choi,
Organic & Biomolecular Chemistry, 2022, 20, 613-618

15. Promotion of 11/9-helical folding in α/β-peptides containing β2-homoalanine residue
Hyerim Yoon, Jaeyeon Lee, Philjae Kang, and Soo Hyuk Choi,
Peptide Science, 2021, e24244.

Sunglim Choi, Ilia A. Guzei, Younghun Kim, Philjae Kang, and Soo Hyuk Choi,
ChemPlusChem, 2021, 86(8), 1069–1073

Karl N. Blodgett, Geunhyuk Jang, Sojung Kim, Min Kyung Kim, Soo Hyuk Choi, and Timothy S. Zwier
Journal of Physical Chemistry A, 2020, 124(28), 5856–5870

Karl N. Blodgett, Joshua L. Fischer, Jaeyeon Lee, Soo Hyuk Choi, and Timothy S. Zwier,
Journal of Physical Chemistry A, 2018, 122(44), 8762-8775
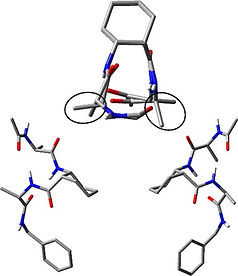
Karl N. Blodgett, Xiao Zhu, Patrick S. Walsh, Dewei Sun, Jaeyeon Lee, Soo Hyuk Choi, and Timothy S. Zwier,
Journal of Physical Chemistry A, 2018, 122(14), 3697-3710
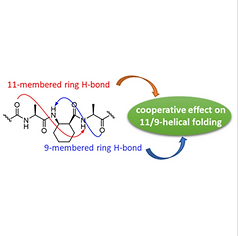
10. Cooperative Effect of the Two Hydrogen Bonding Types on 11/9-Helical Folding of α/β-Peptides
Geunhyuk Jang, Mihye Lee, Jaeyeon Lee, Jihyun Shim, Philjae Kang, Moon-Gun Choi, and Soo Hyuk Choi,
Bulletin of the Korean Chemical Society, 2018, 39(2), 244-249
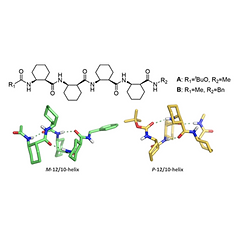
9. Crystal Packing-induced Dimorphism of 12/10-Helical β-Peptides
with Dynamic Folding Propensity
Geunhyuk Jang, Seonho Shin, Ilia A. Guzei, Sooyong, Moon-Gun Choi, and Soo Hyuk Choi,
Bulletin of the Korean Chemical Society, 2018, 39(2), 265-268
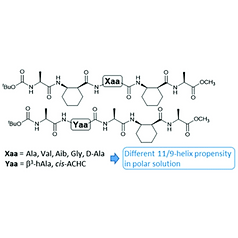
8. Side chain-specific 11/9-helix propensity of α/β-peptides with alternating residue types
Jaeyeon Lee, Jihyun Shim, Philjae Kang, Moon-Gun Choi, and Soo Hyuk Choi, Organic & Biomolecular Chemistry, 2018, 16(3), 433-438
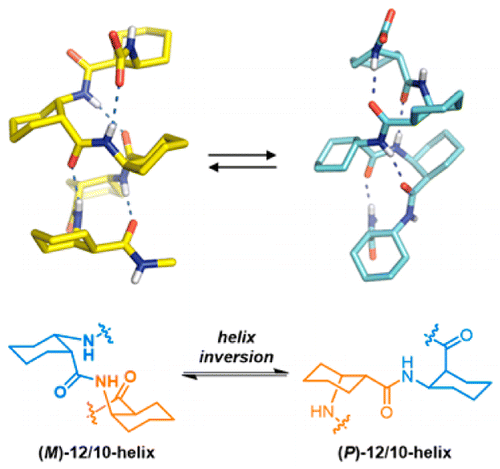
Seonho Shin, Mihye Lee, Ilia A. Guzei, Young Kee Kang, and Soo Hyuk Choi, Journal of American Chemical Society, 2016, 138 (40), 13390–13395

Jaeyeon Lee, Geunhyeok Jang, Philjae Kang, Moon-Gun Choi, and Soo Hyuk Choi, Organic & Biomolecular Chemistry, 2016, 14(36), 8438-8442

5. Stabilization of 11/9-helical α/β-peptide foldamers in protic solvents
Mihye Lee, Jihyun Shim, Philjae Kang, Moon-Gun Choi, and Soo Hyuk Choi, Chemical Communications, 2016, 52(35), 5950-5952

4. cis-2-Aminocyclohex-4-enecarboxylic acid as a new building block of helical foldamers
Sunmi Kwon, Philjae Kang, Moon-Gun Choi, and Soo Hyuk Choi,
New Journal of Chemistry, 2015, 39(5), 3221-3224

3. Helical folding of α/β-peptides containing β-amino acids with an eight-membered ring constraint
Woohyung Lee, Sunmi Kwon, Philjae Kang, Ilia A. Guzei, and Soo Hyuk Choi,
Organic & Biomolecular Chemistry, 2014, 12(17), 2641-2644

2. Helical structures of unnatural peptides for biological applications
Soo Hyuk Choi, Biomedical Engineering Letters, 2013, 3(4), 226-231

Mihye Lee, Jihyun Shim, Philjae Kang, Ilia A Guzei, and Soo Hyuk Choi,
Angewandte Chemie International Edition, 2013, 52(40), 12564-12567